DEPARTMENT OF DEVELOPMENTAL PHYSIOLOGY
We are investigating the neural systems controlling saccadic eye movements and grasping hand movements. We analyze the structure of local circuits and large-scaled networks involved in these motor systems and neural coding of behavioral parameters in these systems. We are also interested in plastic compensatory mechanism following lesion of related structures as detailed below;
1. Saccadic eye movements;
Saccades are rapid eye movements that we execute to capture a visual target in space. Reaction times, trajectories and endpoints of saccades are strongly influenced by attentional state. We investigate the modulatory mechanism of the saccade generator circuits by cognitive functions such as attention by combining multidisciplinary approaches as summarized below. Especially our interest is focused on the superior colliculus (SC), a midbrain structure that plays a key role in generation of saccades.
- Electrophysiological analysis of local circuits of the SC by using in vitro slice preparation. We are investigating the interaction among different layers of the SC, neural mechanism for generation of high frequency presaccadic burst activities, role of GABAergic inhibition, role of NMDA receptors and modulation by cholinergic inputs.
- Analysis of local circuit of SC and large scaled network involving the cerebral cortex for saccade generation in anesthetized animals (rodents and non-human primates).
- Molecular mechanism of saccade generator circuits by using genetically manipulated mice.
- Analysis of dynamic properties of saccade-related circuits by applying electrophysiological and pharmacological techniques in awake behaving non-human primates.
- Clarifying the function of the “extrageniculate visual pathway” by analyzing the performance of visually guided saccades in non-human primates in which the primary visual cortex is unilaterally removed.
2. Dexterous hand movements
Ability to control movements of individual fingers is uniquely developed in primates. It has been supposed that individual finger movements depend on direct connection of corticospinal neurons with motoneurons of intrinsic hand muscles, which appears first in primates phylogenetically. However, recently we clarified the existence of oligosynaptic (indirect) cortico-motoneuronal pathways mediated by interneurons in the cervical spinal cord. Moreover, we observed the behavior of the monkeys in which direct cortico-motoneuronal connections are transected while the indirect pathway remained intact and found that the monkeys can perform precision grip after 1-3 weeks of recovery period. This result indicates that the indirect cortico- motoneuronal pathways can compensate the performance of precision grip to a great deal, which would open up the possibility of neuro-rehabilitation. Furthermore, we think that such compensatory process is not caused simply by the plastic mechanism in the spinal cord but also would involve plastic change in the large-scaled networks involving the cerebral cortex and cerebellum, which may share some common mechanism with learning. To explore the basic mechanism involved in the compensatory mechanisms, we use multidisciplinary approaches including electrophysiology, non-invasive brain imaging with positron emission tomography (PET) and analysis of gene expression by DNA microarray and in situ hybridization.

Figure 1. Precision grip before (A) and after (B) the complete lesion of the lateral corticospinal tract at C5.
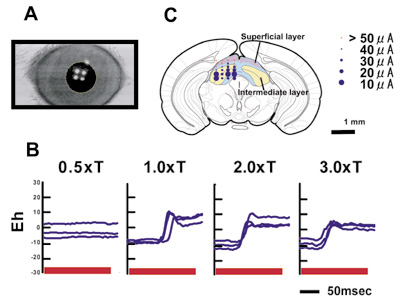
Figure 2. Saccadic eye movements of mice.
A. We developed a high speed on-line videooculography which detects border of cornea with 240 Hz time resolution. B. When we apply high frequency repetitive stimulation to the SC, saccade-like rapid movements are induced. C. The threshold for induction of saccades was low in the deeper layer of the SC.
Staff
 |
Professor:
ISA, Tadashi, MD, PhD
1985 Graduated from University of Tokyo, Faculty of Medicine. 1989 Completed the doctoral course in Science in University of Tokyo. 1989 Research Associate in University of Tokyo. 1993 Lecturer in Gunma University, School of Medicine. 1996 Professor, NIPS.
Speciality: Neurophysiology |
 |
Assistant Professor:
SEKI, Kazuhiko, PhD
1988 Graduated from Niigata University, Faculty of Education. 1998 Completed the doctoral course in Tsukuba University, Faculty of Medicine. 1998 Lecturer in International Budo University, Postdoctoral fellow in University of Washington (Seattle). 2001 Research Associate, NIPS.
Speciality: Neuroscience |
 |
Assistant Professor:
ENDO, Toshiaki, PhD
1996 Graduated from Faculty of Engineering Science, Osaka University. 2000 Completed the doctoral course in Life Science in the Graduate University for Advanced Sciences. 2000 Research Fellow, NIPS. 2002 Research Associate, NIPS.
Speciality: Neuroscience |
 |
Assistant Professor:
YOSHIDA, Masatoshi, PhD
1992 Graduated from University of Tokyo, Department of Pharmaceutical Sciences.1996 Predoctoral Fellow at NIPS and University of Tokyo. 2003 Obtained PhD from University of Tokyo. 2003 Postdoctoral Fellow at University of Tokyo. 2003 Research Associate, NIPS.
Speciallity: Cognitive Neuroscience and Neurophysiology |
 |
Postdoctral Fellow:
SAKATANI, Tomoya, PhD
1997 Graduated from the University of Tokyo, Faculty of Science. 1999 Completed the master course in Biophysics and Biochemistry at the University of Tokyo, Graduate School of Science. 2004 Completed the doctoral course in Physiological Sciences at the Graduate University for Advanced Studies, School of Life Science. 2004 Postdoctral Fellow, NIPS.
Speciality: Neuroscience |
 |
Research Fellow:
NISHIMURA, Yukio, PhD
1995 Graduated from Nihon University, Faculty of Science. 2003 Completed the doctoral course in University of Chiba, Faculty of Humanity and Medicine. 2003 Postdoctral Fellow, NIPS.
Speciality: Neuroscience |
In the last stage of neural development, a massive re-arrangement of neuronal circuits takes place. This is associated with an alteration of neuronal circuits which is already functioned, resulting in the changes in various brain functions e.g. behavior, sensory function and biological rhythm. Our research aim is to understand the developmental re-arrangement of brains function at neuronal circuits and synaptic levels by using various electrophysiological methods and molecular approach. In addition, their modulation by the neural activity driven by intrinsic factors and environments is also studied.
Another question is whether the neural rearragement could re-apper during the recovery after various brain damages, e.g.brain ischemia and injury.
(1) One of our recent targets is to elucidate the developmental plasticity of GABAergic system. In immature animals, GABA induces neuronal depolarization and often acts as a excitatory substance. We study on the cellular and molecular mechanisms of the switch of GABA action from depolarization to hyperpolarization during development. Intracellular CI- concentration decreases during development, resulted from developmental switch of intracellular CI- regulators, e.g. K+ CI- cotransporter2(KCC2) and Na+,K+,CI- cotranasporter 1 (NKCC1). In addition, immediately after neuronal damages, KCC2 rapidly down-regulated in their expression and function, resulting in GABA-induced excitation in injured neurons.
(2)We recently reported a new form of synapse development. Transmitters to the lateral olive neurons, auditory relay neurons, switch from GABA in the immature to glycine in the mature. The transmitter switch proceeds with single synaptic terminals. This could be one of unique synaptic plasticity in developing neural circuits. We now focus on elucidating underlying mechanisms, e.g.related trophic factors.
(3) We also investigate the rapid action of BDNF on GABAergic system of developing visual cortex and hippocampus of rat/ mouse. BDNF rapidly upregulates the surface GABA-A receptor and induces long term enhancement of GABA-induced currents in amplitude. BDNF action switches to rapid down-regulates surface GABA-A receptor in the mature. Underlying mechanisms, including neural activity and GABA-A receptor associated protein, is now under investigation.
(4) In addition, we are interested in elucidating the underlying mechanisms for re-acquisition of cellular and functional immature characteristics in the process of recovery after the cell damage, e.g. GABA excitation resulting from rapid down-regulation of KCC2 expression after neuronal injury, and dominant expression of immature NMDA receptor subunits associated with the alteration of its function.
Staff
 |
Professor:
Nabekura, Junichi, MD, PhD
1980 Graduated from Kyushu University, School of Medicine. 1986 Completed the doctoral course in Medical Sciences, Kyushu University. 1986 Research Fellow, Washington University. 1991 Assistant Professor, Department of Neurophysiology, School of Medicine, Tohoku University. 1993 Associate Professor, Department of Physiology, School of Medicine, Akita University. 1995 Associate Professor, Kyushu University, Graduate School of Medical Sciences. 2003 Professor, NIPS.
Speciality: Neuroscience |
 |
Associate Professor:
JANG, IL-SUNG, PhD
1994 Graduated from Kyunghee University, Faculty of Pharmacy. 1996 Completed the master course in Microbiology, Kyunghee University. 2002 Completed the doctor course in Physiology, Kyushu University. 2004 JSPS postdoctoral fellow for foreign researcher.
Speciality: Neurophysiology |
 |
Assistant Professor:
MAEJIMA, Takashi, PhD
1996 Graduated from Osaka University, Faculty of Engineering Science. 2002 Completed the doctoral course in Medical Science, Kanazawa University. 2002 JSPS Postdoctoral Fellow. 2004 Assistant Professor, NIPS.
Speciality: Neurophysiology |
 |
Research Fellow:
WATANABE, Miho, PhD
1996 Graduated from Waseda University, Faculty of Human Sciences. 1998 Completed the master course in Human Sciences, Waseda University. 2004 Completed the doctoral course in Medicine, Nippon Medical School. 2004 Research Fellow, NIPS.
Speciality: Neuroendocrinology |
 |
JST Research Fellow:
KITAMURA, Akihiko, PhD
1997 Graduated from Hiroshima University, Faculty of Sciences. 1999 Completed the master course in Sciences, Hiroshima University. 2003 JST Research fellow. 2004 Assistant Prfessor, Shinshu University school of Medicine. 2004 Obtained PhD from Osaka University, Medical Sciences. 2004 JST Research Fellow.
Speciality: Neurophysiology |
The animal body has an integrated-regulatory system for “homeostasis”that maintain a normal, constant-internal state by responding to changes in both the external and internal environments. Within the central nervous system, the hypothalamus is a critical center that regulates these homeostatic activities by integrating autonomic nervous system, endocrine system and immune function. In this division, we are extensively investigating the role of hypothalamus in body energy balance in mammals. These studies are now important for better understanding the molecular mechanisms behind pathophysiology of obesity and diabetes mellitus. The main subjects of our current research are as follows;
(1) Molecular mechanism of the hypothalamic regulation of food intake.
(2) Regulatory role of the hypothalamic-sympathetic nervous system in glucose and lipid metabolism.
(3) Signaling mechanism in the hypothalamus that integrates immune reaction, body temperature regulation and feeding behavior.
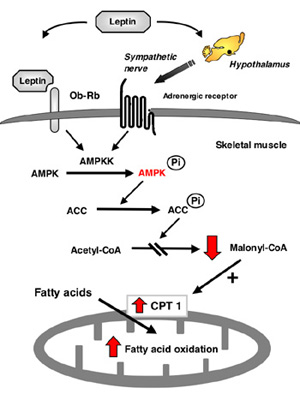
Regulatory mechanism of letpin-induced fatty acid oxidation in muscle.
Leptin activates AMP kinase kinase (AMPKK)/AMP kinase (AMPK) in muscle directly through the leptin receptor Ob-Rb in muscle and indirectly through the hypothalamic-sympathetic nervous system. Activated AMP kinase phosphorylates acetyl-CoA carboxylase (ACC) and inhibits the activity, which decreases malonyl-CoA content. Decrease of malonyl-CoA content stimulates fatty acid oxidation by activating carnitine-palmitoyl transferase 1 (CPT1) in mitochondria.
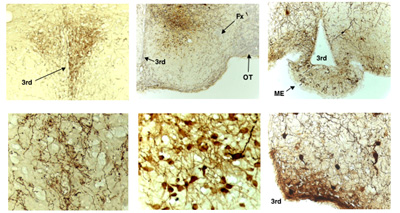
Expression of mutated AMP kinase in the hypothalamus
To explore the role of hypothalamic AMP kinase in body energy balance, we injected adenovirus expressing dominant negative AMP kinase or constitutively-active AMP kinase into the medial hypothalamus. The photographs are immunohistochemistry with a specific antibody against enhanced GFP encoded in the AMP kinase-adenovirus. Left-upper: low magnification of paraventricular hypothalamus (PVH), Left-lower: high magnification of PVH, Middle-upper: low magnification of medial and lateral hypothalamus, Middle-lower: high magnification of ventromedial hypothalamus (VMH), Right-upper: low magnification of arcuate hypothalamus (ARH), Right-lower: high magnification of ARH. 3rd: third ventricle; ME: median eminence; Fx: fornix; OT: optic tract
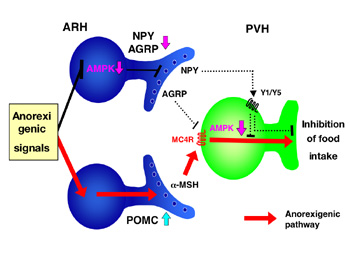
Regulatory role of AMP kinase in the arcuate and paraventricular hypothalamus in food intake
Arcuate hypothalamus (ARH) expresses both POMC anorexigenic and NPY/AGRP orexigenic neurons. POMC anorexigenic neurons releasea-MSH and activate melanocortin 4 receptor (MC4R) in paraventricular hypothalamus (PVH). In contrast, NPY/AGRP neurons release both NPY and AGRP; AGRP inhibits MC4R competitively, and NPY inhibits down-stream signal of MC4R. Anorexigenic signals including leptin inhibits NPY/AGRP neurons by decreasing AMP kinase activity in ARH and activates MC4R signaling in PVH. Activated MC4R also decreases AMP kinase activity in PVH. The suppression of AMP kinase in the hypothalamus is necessary for leptin-induced anorexic effect.
Staff
 |
Professor:
MINOKOSHI, Yasuhiko, MD, PhD
1983 Graduated from Ehime University School of Medicine. 1987 Completed the doctoral course in Science in Ehime University. 1987 Research Associate in Ehime University. 1993 Lecturer in Ehime University School of Medicine. 1997 Associate Professor in Ehime University School of Medicine. 2003 Lecturer in Harvard Medical School. 2003 Professor, NIPS.
Speciality: Endocrinology and metabolism |
 |
Assistant Professor:
SHIUCHI, Tetsuya, PhD
1997 Graduated from Faculty of Integrated Arts and Sciences, The University of Tokushima. 1999 Completed the master course in Graduate School of Human and Natural Environment Sciences, The University of Tokushima. 2003 Completed the doctoral course in Ehime University School of Medicine. 2003 Research Associate, Ehime University School of Medicine. 2004 Research Associate, NIPS.
Speciality: Endocrinology and metabolism, Biochemical physiology of exercise |
 |
Assistant Professor:
OKAMOTO, Shiki, VMD, PhD
1996 Graduated from Faculty of Veterinary Medicine. Hokkaido University, 2000 Completed the doctoral course in Veterinary Medicine in Hokkaido University. 2000 JSPS Research Fellow. 2001 Researcher in Tokyo Metropolitan Institute of Medical Science. 2004 Assistant Professor, NIPS.
Speciality: Neuroimmunology,Stem cell biology |
 |
Postdoctoral Fellow:
LEE, Suni, PhD
1997 Graduated Tokyo University of Agriculture and Technology. 1999 Completed the master course in Graduate School of Tokyo University of Agriculture and Technology. 2002 Completed the PhD course in United Graduate School of Agricultural Sciences, Tokyo University of Agriculture and Technology. 2002 Postdoctoral fellow in Dept. Neurosciences, Case Western Reserve University (OH, US), 2003 Postdoctoral fellow in Dept. Neurological surgery, University of Miami (FL, US). 2005 Postdoctoral fellow, NIPS.
Speciality: Neurocellbiology, Neurosciences |
We are currently examining the neural mechanisms of olfactory learning in three specialized contexts that occur during sensitive periods of enhanced neural plasticity.
(1) Learning of male pheromonal signals by female mice
Male pheromones in mice usually induce oestrus in grouped females and pregnancy block in newly-mated females by initiating a neuroendocrine reflex that suppresses the secretion of prolactin and subsequent progesterone. When a female is mated with a male partner, she forms a memory to the urinary pheromones of the male, thereby preventing his pheromones from inducing pregnancy block. We have demonstrated that the neural changes underlying this memory occur in the accessory olfactory bulb (AOB), the first relay in the vomeronasal system, depend upon mating-induced enhancement of alpha2-adrenergic transmission in the AOB and involve changes at the reciprocal synapses between mitral and granule cells. We are examining the memory mechanisms in greater detail using behavioural pharmacological, neurophysiological and molecular biological approaches.
(2) Olfactory learning in young rats
Rats are altricial and pups are confined to the nest environment with the main sensory input mediated by the olfactory and somatosensory modalities. Pups, however, must learn the odour of the mother to ensure nipple attachment and orientation to the mother. In fact, there is a developmentally determined sensitive period, during which olfactory learning is enhanced. On postnatal day 12, young rats show an aversion to an odour to which they had been exposed along with presentations of foot shock on postnatal day 11. We have indicated that the mitogen-activated protein kinase/extracellular-signal regulated kinase (MAPK/ ERK)-cyclic AMP response element-binding protein (CREB) phosphorylation pathway is required for the long-term, but not the short-term, facilitation process of aversive olfactory learning in young rats. This learning paradigm is particularly appropriate for pharmacological studies in freely behaving animals. We are examining the cellular mechanisms and also approaching the issue of whether long-term potentiation (LTP) is the memory mechanism for aversive olfactory learning in the olfactory bulb.
(3) Maternal memory in rats
Intracerebroventricular injections of oxytocin (OT) induce full maternal behaviour in virgin rats without maternal experience. This observation is of particular importance because virgin rats find pup odours aversive and, thus far, no other peptide has induced a rat to overcome this aversion. Our studies have previously demonstrated that the olfactory bulb is a critical site where OT acts to induce a rapid onset of maternal behaviour. Our studies have further demonstrated that OT not only modulates synaptic transmission at the reciprocal synapses between mitral and granule cells but also induces LTP at these synapses. We are investigating the cellular mechanism for OT-induced LTP.
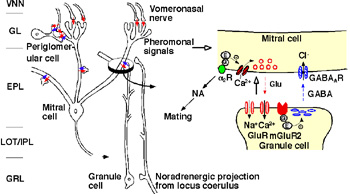
Figure 1. Major synaptic interconnection in the AOB and a cellular mechanism for pheromonal memory formation
Staff
 |
Professor:
KABA, Hideto, DVM, PhD
1974 Graduated from Kagoshima University, Faculty of Agriculture.
1976 Completed the master course in Agriculture, Kagoshima University. 1982 Completed the doctoral course in Nutrition, University of Tokushima. 1980 Instructor and 1988 Associate Professor, Kochi Medical School. 1995 Professor, Kagoshima University, Faculty of Agriculture. 1997 Professor, Kochi Medical School. 2003 Adjunct Professor. NIPS.
Speciality: Neuroscience. |
|





















