DEPARTMENT OF CELL PHYSIOLOGY
Outline
Department of Cell Physiology is composed of four divisions, Division of Membrane Physiology, Correlative Physiology, Neural Systematics, and Cell Signaling. Divisions of Neural Systematics and Cell Signaling are adjunct and joint divisions, respectively. To clarify roles of channels, transporters, receptors, biosensor channels (TRPs), enzymes and their protein complexes in physiological functions, researches are being done mainly at the levels ranging from the molecule, cell, and tissue to organ. In addition, researches are being conducted to elucidate the pathological mechanisms associated with their dysfunction. Furthermore, studies on regulatory mechanisms for processing visual information in retina and sleep/wakefulness are being done.
Each division has the latest research technology and methods on molecular and cellular biology, biochemistry and proteomics, electrophysiology, neuroanatomy, and molecular genetics. Sharing and developing these technology and methods, we are attempting to reveal the physiological function of human body in an integrative manner.
Research focuses on membrane proteins and especially on the functional proteins localized in the synaptic membranes of the brain (neurotransmitter receptors, ion channels, adhesion molecules, and so on). Synaptic membrane proteins do not function alone, but form protein complexes (functional units) with anchoring proteins, signaling proteins, and thereby performing their physiological functions. Many synaptic proteins undergo protein palmitoylation, and the localization on the synaptic membrane is dynamically regulated by neuronal activity. The goal is to elucidate the molecular mechanism determining the dynamic localization for synaptic proteins, by the functional analyses of the palmitoylating enzymes we recently discovered. In addition, taking advantage of our unique biochemical techniques, we are purifying and identifying novel synaptic protein complexes from the brain tissue, in particular focusing on disease candidate proteins. Based on these molecular clues, we will elucidate the molecular mechanism for controlling synaptic transmission by multidisciplinary approaches.
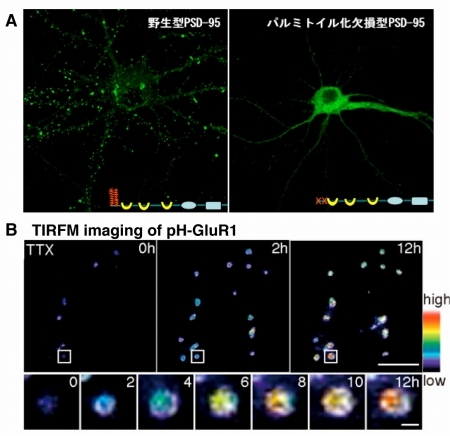
Figure 1. Palmitoylation determines the localization of various synaptic proteins.
Protein palmitoylation is a common posttranslational lipid modification and regulates the membrane targeting of proteins such as a postsynaptic scaffold, PSD-95. Palmitoylation is unique in that it is reversible and dynamically regulated by specific extracellular signals. The reversible nature of protein palmitoylation allows proteins to shuttle between intracellular compartments. (A) For example, wild-type PSD-95 clearly targets to postsynaptic membrane in hippocampal neurons (left), whereas palmitoylation deficient mutant of PSD-95 diffusely localizes in somato-dendrites (right). (B) Upon activity blockade, a palmitoylation enzyme translocates to postsynaptic density and induces PSD-95 palmitoylation and mediates synaptic clustering of PSD-95 and associated AMPA-type glutamate receprotors. We are currently challenging the question how dynamic cycling between palmitoylation and depalmitoylation is regulated inside living neurons.
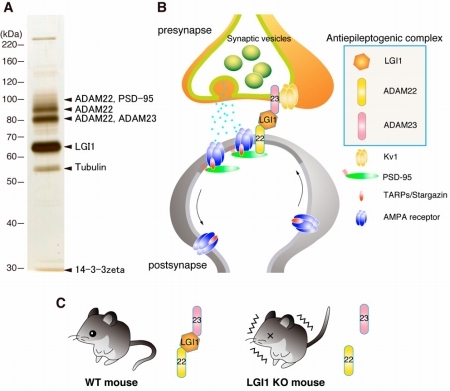
Figure 2. Synaptic proteins do not usually function alone, but form protein complexes with anchoring proteins and signaling proteins, thereby performing their physiological functions. Taking advantage of our unique biochemical approaches, we have identified various physiological protein complexes. Examples include PSD-95- and LGI1-containing protein complexes. LGI1 is a neuronal secreted protein whose mutations were reported in patients with an inherited form of human epilepsy. We recently purified the LGI1-containing protein complexes from mouse brain, and found that secreted protein LGI1 functions as a ligand for ADAM22 and ADAM23. Interestingly loss of LGI1, ADAM22, or ADAM23 in mice showed similar epileptic seizures. Also, loss of LGI1 reduces AMPA receptor-mediated synaptic transmission.
Thus, we will clarify the major protein-protein networks and the various physiological functions, such as synaptic transmission in the brain.
Staff
FUKATA, Masaki, MD, PhD
1994 Graduated from Kobe University School of Medicine. 2000 Completed the doctoral course in Medical Sciences, Hiroshima University. 2000 JSPS Research Fellow. 2001 Assistant Professor, Nagoya University. 2003 JSPS Postdoctoral Fellow for Research Abroad, University of California at San Francisco. 2005 Section Chief, National Center for Geriatrics and Gerontology. 2007 Professor, NIPS.
Speciality: Neuroscience, Biochemistry, Cell biology
FUKATA, Yuko, MD, PhD
1994 Graduated from Kobe University School of Medicine. 2000 Completed the doctoral course in Bioscience, Nara Institute of Science and Technology. 2000 JSPS Research Fellow, Nagoya University. 2003 HFSP Long-term fellow, University of California at San Francisco. 2005 Research fellow, National Center for Geriatrics and Gerontology. 2007 Research fellow, NIPS. 2007 PRESTO Researcher, JST. 2007 Associate Professor, NIPS.
Speciality: Neuroscience, Biochemistry, Cell biology
IWANAGA, Tsuyoshi, PhD
1993 Graduated from College of Mining, Akita University. 1995 Completed the master course in Department of Materials and Applied chemistry, Akita University. 2001 Completed the doctor course in Faculty of Integrated Arts and Sciences, Hiroshima University. 2001 Research Fellow, National Cardiovascular Center. 2005 Research Associate, Kyoto University. 2006 Research Fellow, National Center for Geriatrics and Gerontology. 2008 Project Assistant Professor, NIPS.
Speciality: Molecular biology, Biochemistry
NORITAKE, Jun, PhD
2000 Graduated from College of Art and Science, University of Massachusetts. 2003 Completed the master course in Graduate School of Medicine, Nagoya University. 2006 Completed the doctoral course in Graduate School of Medicine, Nagoya University. 2006 JSPS Postdoctoral Research Fellow, Nagoya University. 2007 Postdoctoral JSPS Research Fellow, NIPS, 2010 Postdoctoral fellow, NIPS.
Speciality: Neuroscience, Cell biology
YOKOI, Norihiko, PhD
2003 Graduated from Faculty of Science, Nagoya University. 2005 Completed the master course in Graduate School of Science, Nagoya University. 2009 Completed the doctoral course in Graduate School of Science, Nagoya University. 2009 Postdoctoral Fellow, NIPS. 2010 JSPS Postdoctoral Fellow, NIPS.
Speciality: Bioinorganic chemistry, Organic synthetic chemistry, Protein crystallography
All of the cell functions, including neural activities, are performed or supported by operation of bio-molecular sensors, channels (ion and water channels) and transporters (carriers and pumps) located on the membrane. The objectives of our division work are to elucidate molecular mechanisms of most general cell activities, such as volume regulation, absorption/secretion and environmental signal reception, to clarify roles of channels, transporters and receptors in these fundamental functions from the viewpoint of integrative biology, and to throw the light on the relationship between these malfunctions and diseases or cell death, as well as to study the multifunctionality of channel and transporter during cell functions or malfunctions.
The main subjects of our current research are as follows:
(1) "Mechanisms of processing visual information in retina" In the retina, various kinds of bio-molecular sensors express in retinal neurons to form early vision. We investigate visual function in retina, especially visual integration mechanisms in retinal ganglion cells by gene-manipulation of bio-molecular sensors and electrophysiology with organotypic retinal culture system (Fig. 1).
(2) "Molecular mechanisms of cell volume regulation and their physiological roles": Most cells regulate their cell volume even under anisotonic conditions. In the volume regulation mechanisms, a number of channels, transporters and receptors are involved (Fig. 2). We are investigating to identify volume-regulatory membrane machineries, including the volume-sensitive anion channel, and to clarify their physiological roles.
(3) "Induction mechanisms of apoptotic, necrotic and ischemic cell death": Dysfunction of cell volume regulation is associated with necrotic and apoptotic cell death (Fig. 3) which is coupled to persistent swelling (necrotic volume increase: NVI) and shrinkage (apoptotic volume decrease: AVD). Our aim is to pioneer the new field of 'PHYSIOLOGY OF CELL DEATH' through elucidation of the mechanisms of cell volume regulation and their dysfunction. We are attempting to focus our studies on the mechanisms of ischemic cell death of brain neurons and cardiac myocytes.
(4) "Molecular mechanisms of channel functions as bio-molecular sensors": Channels are multifunctional proteins involved not only in electric signal generation and ion transport but also in sensing the environmental factors or stress. We aim at elucidating molecular mechanisms of volume- and stress-sensing functions of anion channels, ATP channels and TRP cation channels (Fig. 4).
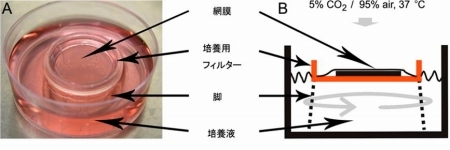
Fig. 1 Oraganotypic culture of adult retina and gene transfection [after Koizumi et al, 2007, PLoS One]
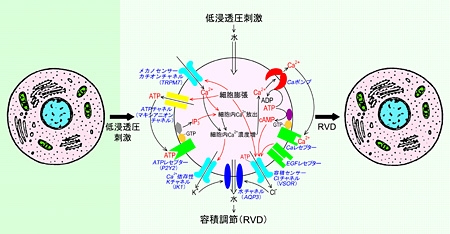
Fig. 2 Molecular mechanisms of the regulatory volume decrease (RVD) and of volume-sensor Cl channel (VSOR) activation. [after Okada et al. 2001, J. Physiol. 532, 3-16]
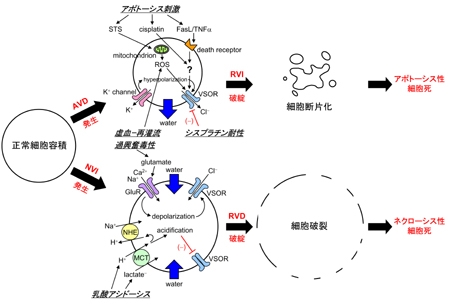
Fig. 3 Roles of channels and transporters in the induction of apoptotic volume decrease (AVD) and apoptotic cell death as well as in that of necrotic volume increase (NVI) and necrotic cell death. [after Okada et al. 2009, J Physiol.]
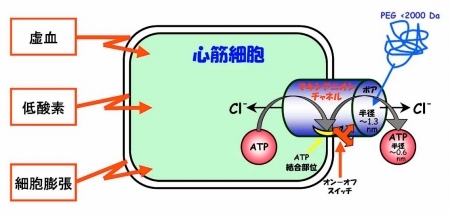
Fig. 4 Stress-sensing, ATP-releasing maxi-anion channel in cardiomyocytes. [after Sabirov & Okada 2005, Purinergic Signalling. 1, 311-328]
Staff
OKADA, Yasunobu, MD, PhD
1970 Graduated from Kyoto University, Faculty of Medicine. 1974 Instructor and 1981 Assistant Professor, Kyoto University, Faculty of Medicine. 1992 Professor, NIPS. 2004 Vice-Director-General. 2007 Director-General, NIPS.
Speciality: Molecular and Cellular Physiology
KOIZUMI, Amane, MD, PhD
1997 Graduated from Keio University School of Medicine. Assistant Professor in Department of Physiology, Keio University School of Medicine. 2002 PhD in Neurophysiology, Keio University. 2002-2007 Research Associate, in Howard Hughes Medical Institute, Harvard Medical School, and Massachusetts General Hospital. 2007 Associate Professor, NIPS.
Speciality: Vision research, Neurophysiology
OKADA, Toshiaki, PhD
1988 Graduated from Okayama University, Faculty of Science. 1993 Completed the doctoral course in Natural Science and Technology, Okayama University. 1996 post-doc in AIST. 2000 postdoc in State University of New York at Stony Brook. 2002 Research scientist in RIKEN. 2009 Project Associate Professor, NIPS.
Speciality: Developmental Biology, Developmental Neuroscience
KUKITA, Fumio, PhD
1971 Graduated from the University of Tokyo, Faculty of Science. 1976 Completed the doctoral course in physics at the University of Tokyo, 1977 Research Associate, NIPS.
Speciality: Biophysics and Molecular Physiology
KASHIHARA, Yasuhiro, PhD
1976 Graduated from Toyama University, Faculty of Science. 1983 Completed the doctoral course in Science, Kyushu University. 1983 Research Associate, NIPS.
Speciality: Neurobiology
MOHRI, Tatsuma, PhD
1978 Graduated from Yamaguchi University. 1981 Completed a master course in Physics, Kanazawa University. 1991 Completed a doctoral course in Life Chemistry, Tokyo Institute of Technology. 1991 Jean and Katsuma Dan Fellow, Hopkins Marine Station Stanford University. 1991 Postdoctoral Associate and 1993 Research Associate, University of Miami School of Medicine. 1995 Postdoctoral Researcher, University of California Davis. 1996 Research Associate, NIPS.
Specialty: Cell Biology, Cell Physiology
AKITA, Tenpei, MD, PhD
1996 Graduated from Saga Medical School, Institute of Medicine. 2001 Completed the doctoral course in Physiology, Nagoya University Graduate School of Medicine. 2001 JSPS Postdoctoral Fellow. 2004 Research Fellow, Nagoya University of Arts and Sciences. 2008 Research Associate, NIPS.
Speciality: Cell Physiology, Neurophysiology
URAMOTO, Hiromi, PhD
1990 Graduated from Japan Women's University, Department of Food and Nutrition. 2002 Left the doctoral course in Life Science, the Graduate University for Advanced Studies. Nov 2002 JST Research Fellow. Apr 2004 MEXT Postdoctoral Fellow.
Speciality: Cell Physiology
Division of Neural Systematics
Division of Cell Signaling
Department of Bio-Environmental Science,
OKAZAKI INSTITUTE FOR
INTEGRATIVE BIOSCIENCE
We mainly investigate molecular mechanisms of thermosensation, nociception, taste sensation and Ca2+ absorption by focusing on TRP ion channels. We also investigate molecular mechanisms of sleep/wakefulness regulation by focusing on orexin neurons. Molecular cell biological, biochemical, developmental biological and electrophysiological techniques are utilized to achieve the above objectives. The followings are major projects in progress.
(1) Molecular mechanisms of thermosensation: Temperature sensing ability is conferred by ion channels of the TRPV, TRPM and TRPA families. We try to clarify the molecular mechanisms of thermosensation and their physiological significance by focusing on those thermosensitive TRP channels from mammals to insects. We are also doing behavioral analyses of mice lacking TRPV3, TRPV4 or TRPM2. Furthermore, we are trying to isolate a novel thermosensitive TRP channels.
(2) Molecular mechanisms of nociception: Capsaicin receptor TRPV1 and TRPA1 are ion channels activated by different noxious stimuli. We try to clarify the nociceptive mechanisms at peripheral nerve endings by focusing on TRP ion channels, especially TRPV1 and TRPA1. We are also doing behavioral analyses of TRPV1- or TRPA1-deficient mice.
(3) We study neurons in the hypothalamus. The hypothalamus is implicated in the maintenance of homeostasis, such as body temperature regulation, feeding regulation and sleep/wakefulness regulation. We make transgenic animals and analyze them by using many techniques including electrophysiological analysis such as slice patch clamp and in vivo extracellular recording as well as immunohistochemical analysis and behavioral analysis such as sleep recording. We try to reveal the neural mechanism, which involved in the maintenance of homeostasis, in molecular, cellular and whole animal level.
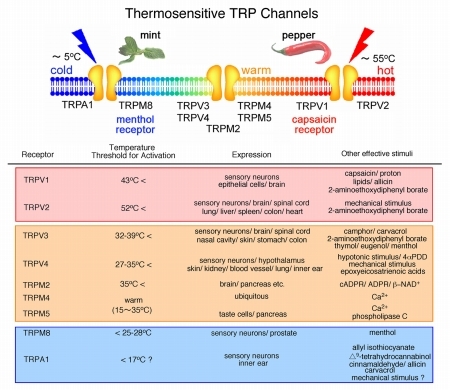
[Mammalian thermosensitive TRP channels (upper) and their properties (lower)]
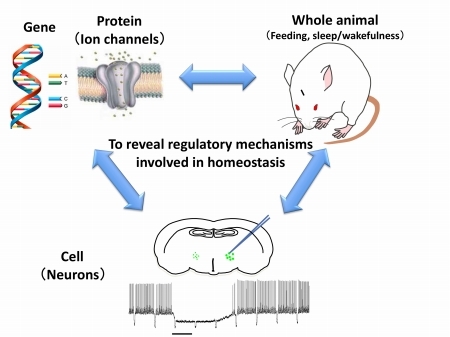
[Studying the regulatory mechanisms involved in feeding and sleep/wakefulness at multi levels: molecular, cellular and whole animal levels]
Staff
TOMINAGA, Makoto, MD, PhD
1984 Graduated from Ehime University, Faculty of Medicine. 1992 Graduated from Kyoto University Graduate School of Medicine. 1993-1999 Assistant Professor, National Institute for Physiological Sciences. 1996-1999 Research Fellow, University of California at San Francisco. 1999-2000 Assistant Professor, University of Tsukuba. 2000-2004 Professor, Mie University School of Medicine. 2004 Professor, Okazaki Institute for Integrative Bioscience.
Specialty: Cellular and Molecular Physiology
YAMANAKA, Akihiro, PhD
1994 Graduated from University of Shizuoka, Faculty of Pharmacy. 1994-1996 University of Tsukuba, 1996-2000 University of Tsukuba. 2000-2002 Researcher, Institute of Basic Medical Sciences, University of Tsukuba. 2002-2008 Assistant Professor, Institute of Basic Medical Sciences, University of Tsukuba. 2006-2008 Research Fellow in Abroad, JSPS. 2008 Associate Professor, Okazaki Institute for Integrative Bioscience.
Specialty: Pharmacology, Neuroscience
SUZUKI, Yoshiro, PhD
2000 Graduated from Tokyo Institute of Technology, Graduate School of Bioscience and Biotechnology. 2000-2002 JSPS Research Fellow, 2002 Research Associate, Kitasato University School of Medicine. 2003 Research Fellow, Brigham and Women's Hospital/Harvard Medical School, 2005 JSPS Research Fellow in Abroad (University of Bern, Switzerland), 2009 Senior Research Fellow, University of Bern, 2010 Assistant Professor, Okazaki Institute for Integrative Bioscience.
Specialty: Molecular Biology, Nephrology
SOKABE, Takaaki, PhD
1998 Graduated from Himeji Institute of Technology, Faculty of Science. 2004 Graduated from University of Tokyo Graduate School of Medicine. 2004-2005 Research Fellow, University of Tokyo Graduate School of Medicine. 2005 Postdoctoral Fellow, National Institute for Physiological Sciences. 2007 JSPS Postdoctoral Fellow. 2008 Assistant Professor, National Institute for Physiological Sciences.
Specialty: Cellular and Molecular Biology
BOUDAKA, Ammar, MD, PhD
1998 Graduated from Al-Fateh University, Faculty of Medicine, Tripoli, Libya. 1998-2002 General Practitioner at Al-Fateh Clinic, Tripoli, Libya. 2007 Graduated from Gifu University, The United Graduate School. 2007-2008 Postdoctoral Fellow, University of Erlangen-Nuremberg, Institute of Anatomy, Germany. 2008 JSPS Postdoctoral Fellow, National Institute for Physiological Sciences.
Specialty: Cellular and Molecular Physiology
SAITO, Shigeru, PhD
1997 Graduated from Hirosaki University, Faculty of Sciences. 1999 Graduated from Tokyo Metropolitan University, Graduate School of Sciences. 2004 Graduated from Tokyo Metropolitan University, Graduate School of Sciences. 2004-2005 Postdoctoral Fellow, Faculty of Agriculture, Iwate University. 2005-2009 Postdoctoral Fellow, 21st Century COE Program, Iwate University. 2009 Postdoctoral Fellow, Okazaki Institute for Integrative Bioscience.
Specialty: Molecular evolution, Comparative genomics
UMEMURA, Toru, PhD
1994 Graduated from Nagoya University, Faculty of Science. 2000 Graduated from Nagoya University, Graduate School of Science. 2000-2000 Postdoctoral Fellow, Nagoya University. 2000-2004 Postdoctoral Fellow, Waksman Institute, Rutgers University. 2004-2008 Postdoctoral Fellow, National Institute of Genetics. 2008 Postdoctoral Fellow National Institute for Physiological Sciences.
Specialty: Molecular Biology
UCHIDA, Kunitoshi, PhD
2003 Graduated from Nagoya City University, Faculty of Pharmacy. 2005 Graduated from Graduate School of Pharmaceutical Sciences, Nagoya City University. 2005-2007 Pharmaceutical Research Center, NIPRO Corporation. 2010 Graduated from the Graduate University for Advanced Studies, School of Life Science. 2010 Postdoctoral Fellow, Okazaki Institute for Integrative Bioscience.
Specialty: Cellular and Molecular Physiology




















