Stretch- and swelling-activated cation (SSAC) channels play essential roles not only in sensing and transducing external mechanical stresses but also in regulating cell volume in living cells. However, the molecular nature of SSAC channel has not been clarified. In human epithelial HeLa cells, single-channel recordings in cell-attached and inside-out patches revealed expression of a Mg2+- and Gd3+-sensitive non-selective cation channel which is sensitive to membrane stretch. Whole-cell recordings revealed that the macroscopic cationic currents exhibit TRPM7-like properties and were augmented by osmotic cell swelling. RT-PCR and Western blotting demonstrated molecular expression of TRPM7 in HeLa cells. Treatment with small interfering RNA (siRNA) targeted against TRPM7 led to abolition of single stretch-activated cation channel currents and of swelling-activated, whole-cell cation currents in HeLa cells. The silencing of TRPM7 by siRNA reduced the rate of cell volume recovery after osmotic swelling. In HEK293T cells, whole-cell currents of the TRPM7 channel heterologously expressed were found to be augmented by mechano-stress due not only to perfusion of bath solution but also to osmotic swelling. Furthermore, in cell-free patches, membrane stretch directly augmented single-channel activity of TRPM7 by increasing Po value. It is thus concluded that TRPM7 represents the SSAC channel endogenously expressed in HeLa cells and heterologously expressed in HEK293T cells and that, by serving as a swelling-induced Ca2+ influx pathway, it plays an important role in cell volume regulation.
Numata T, Shimizu T, Okada Y. (2007) TRPM7 is a stretch- and swelling-activated cation channel involved in volume regulation in human epithelial cells. Am J Physiol Cell Physiol 292: C460-467
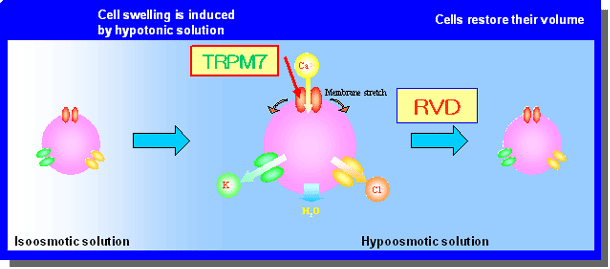
A schematic view of RVD. Cell volume is increased by application of hypoosmotic solution. In response to increased membrane tension, stretch-activated TRPM7 cation channels causes Ca influx. This Ca influx then triggers an increase in intracellular Ca, thereby activating Ca-activated K channels. In parallel, volume expansion activates volume-sensitive outward rectifying Cl channels. The resultant KCl efflux drives osmotically obliged water, and thereby restores their cell volume.