Expression of the TRPM6 in mouse placental trophoblasts; potential role in maternal-fetal calcium transport
The placenta is required to transport minerals such as calcium and magnesium from mother to fetus during fetal bone mineralization. If disturbed, it leads serious symptoms not only in fetal bones but also in the development of fetal brain and cardiovascular system. Previously we have shown that TRPV6 calcium-selective channel is critical for the maternal-fetal calcium transport for fetal bone formation.
In this study, we identified another TRP channel, TRPM6 as another candidate mineral transport pathway in mice. TRPM6 mRNA and proteins localized in trophoblasts where the maternal-fetal mineral transport occurs, and the message increased coinciding with fetal bone mineralization. In whole-cell patch-clamp recordings, we observed endogenous TRPM6/TRPM7-like currents in primary mouse trophoblasts after starting fetal bone mineralization but not before starting.
These suggest that TRPM6 is functionally expressed in mouse placental trophoblasts as TRPM6/TRPM7 heteromers, implicating in the maternal-fetal mineral transport that enable to sustain fetal bone mineralization.
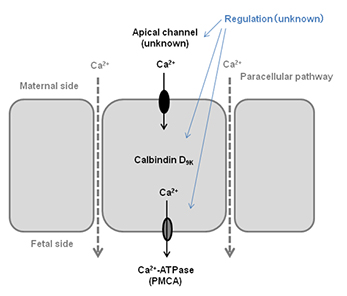
Molecular mechanism of mineral (calcium) transport in the placenta
In the last trimester, calcium is actively transported from mother to fetus via transcellular pathway, which is composed by the apical ion channel, intracellular calcium binding protein (calbindin D), and plasma membrane Ca2+-ATPase (PMCA). TRPV6 and TRPM6 are candidate apical mineral entry channels.
Questions
Q1: What is interesting to you about this topic?
A1: TRPM6 mutations have been reported in familial hypomagnesemic patients, but to our knowledge, there has been no report showing the activity of endogenous TRPM6 channel in tissues. We believe this is the first report showing the endogenous TRPM6-like membrane currents.
Q2: Did something surprise you when you did this study and analyzed your results?
A2: We were surprised that the actual membrane currents were not like TRPM6 homomer’s, but TRPM6/TRPM7 heteromer’s. Since TRPM7 is expressed ubiquitously, we propose that the regulation of TRPM6 gene might be important for total body mineral homeostasis.
Funding
KAKENHI #22890248 and #23790266

