| |
2025
-
Matsushima K, Sumikama T, Suzuki T, Ito M, Nagasawa Y, Sumino A, Flechsig H, Ogoshi T, Umeda K, Kodera N,
Murakoshi H.*, Shibata M.*.
Structural dynamics of mixed-subunit CaMKIIα/β heterododecamers filmed by high-speed AFM.
Nature communications,16:10603 (2025).
2024
-
Anjum R, Clarke VRJ, Nagasawa Y, Murakoshi H, Paradis S*.
Rem2 interacts with CaMKII at synapses and restricts long-term potentiation in hippocampus.
PLoS ONE 19(7), e0301063 (2024).
2023
-
Tsujioka S†, Sumino A†, Nagasawa Y†, Sumikama T, Flechsig H, Puppulin L, Tomita T, Baba Y, Kakuta T, Ogoshi T, Umeda K, Kodera N, Murakoshi H*, Shibata M*.
Imaging single CaMKII holoenzymes at work by high-speed atomic force microscopy.
Science Advances 9(26), eadh1069 (2023).
-
Nagasawa Y, Ueda HH, Kawabata H, and Murakoshi H*.
LOV2-based photoactivatable CaMKII and its application to single synapses: Local Optogenetics.
Biophysics and Physicobiology 20, e200027 (2023).
2022
-
Murakoshi H*, Ueda HH, Goto R, Hamada K, Nagasawa Y, and Fuji T*.
In vivo three- and four-photon fluorescence microscopy using a 1.8 μm femtosecond fiber laser system.
Biomedical Optics Express 14(1), 326-334 (2022).
-
Ueda HH, Nagasawa Y, Sato A, Onda M, and Murakoshi H*.
Chronic neuronal excitation leads to dual metaplasticity in the signaling for structural long-term potentiation.
Cell Reports 38, 110153 (2022).
2021
-
Shimizu T, Murakoshi H, Matsumoto H, Ichino K, Hattori A, Ueno S, Ishida A, Tajiri N, Hida H.
Tension sensor based on fluorescence resonance energy transfer reveals fiber diameter-dependent mechanical factors during myelination.
Frontiers in Cellular Neuroscience 15, 685044 (2021).
-
Shibata AC, Ueda HH, Eto K, Onda M, Sato A, Ohba T, Nabekura J, and Murakoshi H*.
Photoactivatable CaMKII induces synaptic plasticity in single synapses.
Nature communications, 12, 751 (2021).
- Ueda HH, Nagasawa Y, and Murakoshi H*
Imaging intracellular protein interactions/activity in neurons using 2-photon fluorescence lifetime imaging microscopy.
Neuroscience Research, S0168-0102(21)00212-1. (2021).
- Murakoshi H*
Optogenetic imaging of protein activity in the synapse by using 2-photon fluorescence lifetime imaging microscopy.
Optogenetics. Springer-Verlag, the Advances in Experimental Medicine and Biology book series 295–308
(2021).
- 植田大海、長澤裕太郎、村越秀治
「光応答性CaMKIIの開発と単一シナプス光操作 ―ローカルオプトジェネティクス―」
生物物理 2021年11月 p374–377
2020
-
Sato A, Arichi S, Kojima F, Hayashi T, Ohba T, Lawrence Cheung D, Narushima M, Murakoshi H, Maruo Y, Kadoya Y, Nabekura J, Ishibashi H.
Histamine depolarizes rat intracardiac ganglion neurons through the activation of TRPC non-selective cation channels.
European journal of Pharmacology, 886; 173536 (2020)
-
Nomura Y, Murakoshi H, and Fuji T.
Short-wavelength, ultrafast thulium-doped fiber laser system for three-photon microscopy.
OSA continuum, 3(6), 1428–1435 (2020)
- 村越秀治、植田大海
「2光子蛍光寿命イメージングと色素タンパク質によるタンパク質間相互作用の可視化」
光学 2020年 49巻1号 p20–25
2019
-
Murakoshi H*, Horiuchi H, Kosugi T, Onda M, Sato A, Koga N, and Nabekura J.
ShadowR: a novel chromoprotein with reduced non-specific binding and improved expression in living cells.
Scientific Reports 9, 12072 (2019).
-
Saneyoshi T, Matsuno H, Suzuki A, Murakoshi H, Hedrick NG, Agnello E, O'Connell R, Stratton MM, Yasuda R, and Hayashi Y.
Reciprocal Activation within a Kinase-Effector Complex Underlying Persistence
of Structural LTP.
Neuron 102, 1199–1210 (2019)
2018
-
Chen Xi, Shibata AC, Hendi A, Kurashina M., Fortes E, Weilinger N, MacVicar B, Murakoshi H, Mizumoto K.
Rap2 and TNIK control Plexin-dependent tiled synaptic innervation in C.
elegans.
eLife, 7:e38801 (2018).
-
Murakoshi H.
Neuronal Synaptic Connections Organized by Small Numbers of Molecules.
Minorities and Small Numbers from Molecules to Organisms in Biology.
Springer-Verlag, Germany. p7–13 (2018)
- 村越秀治 「共焦点レーザー走査顕微鏡v.FLIM-②周波数ドメイン」
生きてるものは全部観る! イメージングの選び方・使い方100+
原田慶恵,永井健治[編]
実験医学増刊 羊土社 2018年12月 p57–58
-
村越秀治 「光応答性CaMKII阻害ペプチドの開発とシナプス可塑性研究への応用」
CLINICAL CALCIUM 医薬ジャーナル社、2018年 p414–419
2017
-
Murakoshi H*, Shibata AC.
ShadowY: a dark yellow fluorescent protein for FLIM-based FRET measurement.
Scientific Reports 7, 6791 (2017)
-
Murakoshi H*, Shin M, Parra-Bueno P, Szatmari EM, Shibata AC, & Yasuda R.
Kinetics of endogenous CaMKII required for synaptic plasticity revealed by optogenetic kinase inhibitor.
Neuron 94, 37–47 (2017)
-
Nakahata Y, Eto K, Murakoshi H, Watanabe M, Kuriu T, Hirata H, Moorhouse AJ, Ishibashi H, & Nabekura J.
Activation-dependent rapid postsynaptic clustering of glycine receptors in mature spinal cord neurons.
eNeuro 4, 0194–16 (2017)
-
村越秀治 「分子間相互作用を検出する2光子蛍光寿命イメージング」
生体の科学 医学書院、2017年 p400–401
-
村越秀治 「クローズアップ実験法 光応答性阻害ペプチドの生化学的機能アッセイ」
実験医学 羊土社、2017年 p2765–2770
-
村越秀治 「第2章 少数性が担う神経シナプス機能」
少数性生物学 永井健治・冨樫祐一[編] 日本評論社、2017年 p9–16
2016
-
Nakahata Y, Nabekura J, & Murakoshi H*.
Dual observation of the ATP-evoked small GTPase activation and Ca2+ transient in astrocytes using a dark red fluorescent protein.
Scientific Reports 6, 39564 (2016)
-
Phengchat R, Takata H, Morii K, Inada N, Murakoshi H, Uchiyama S, & Fukui K.
Calcium ions function as a booster of chromosome condensation.
Scientific Reports 6, 38281 (2016)
-
Hedrick NG, Harward SC, Hall CE, Murakoshi H, McNamara JO, & Yasuda R.
Rho GTPase complementation underlies BDNF-dependent homo- and heterosynaptic plasticity.
Nature 538, 104–108 (2016)
-
Miyamoto A, Wake H, Ishikawa AW, Eto K, Shibata K, Murakoshi H, KoizumiS, Moorhouse AJ, Yoshimura Y, & Nabekura J.
Microglia contact induces synapse formation in developing somatosensory cortex.
Nature Communications 7, 12540 (2016)
-
Fujiwara TK, Iwasawa K, Kalay Z, Tsunoyama TA, Watanabe Y, Umemura YM,
Murakoshi H, Suzuki KG, Nemoto YL, Morone N, & Kusumi A.
Confined diffusion of transmembrane proteins and lipids induced by the
same actin meshwork lining the plasma membrane.
Molecular Biology of the Cell 27, 1101–1119 (2016)
-
Murakoshi H and Yasuda R.
Imaging signal transduction in dendrites using genetically-encoded fluorescent proteins.
Dendrite -development and disease-
Springer-Verlag, Germany. p139–154 (2016)
-
柴田明裕、村越秀治
「細胞内発現パターンを劇的に改善した高精度FRETセンサーの開発」
生物物理 2016年 56-3 p181–184
2015年以前
原著論文
-
Murakoshi H*, Shibata AC, Nakahata Y, & Nabekura J.
A dark green fluorescent protein as an acceptor for measurement of Förster resonance energy transfer by fluorescence lifetime imaging microscopy.
Scientific Reports 5, 15334 (2015)
-
Shibata AC, Maebashi KH, Nakahata Y, Nabekura J, & Murakoshi H*.
Development of a molecularly evolved, highly sensitive CaMKII FRET sensor with improved expression pattern.
PLoS ONE 10(3), e0121109 (2015)
-
Uezu A,Okada H, Murakoshi H, Del Vescovo CD, Yasuda R, Diviani D, & Soderling S.
A Modified SH2 Domain to Phototrap and Identify Phosphotyrosine Proteins from Subcellular Sites within Cells.
PNAS. 109, E2929- E2938 (2012)
-
Murakoshi H, Wang H, Yasuda R.
Local,persistent activation of Rho GTPases during plasticity of single dendritic spines.
Nature 472, 100–104 (2011)
-
Murakoshi H, Seok-Jin Lee, Yasuda R.
Highly sensitive and quantitative FRET-FLIM imaging in single dendritic spines using improved non-radiative YFP.
Brain Cell Biol. 36, 31–42 (2008)
表紙に採択されました。

-
Koyama-Honda I, Ritchie K, FujiwaraT, Iino R, Murakoshi H, Kasai RS, & Kusumi A.
Fluorescence imaging for monitoring the colocalization of two single molecules in living cells.
Biophys J. 88, 2126–2136 (2005)
-
Murase K, Fujiwara T, Umemura Y, Suzuki K, Iino R,Yamashita H, Saito M,Murakoshi H, Ritchie K, & Kusumi A.
Ultrafine membrane compartments for molecular diffusion as revealed bysingle molecule techniques.
Biophys J. 86, 4075–4093 (2004)
-
Murakoshi H, Iino R, Kobayashi T, Fujiwara T, Ohshima C, Yoshimura A & Kusumi A.
Single-molecule imaging analysis of Ras activation in living cells.
PNAS. 101, 7317–7322 (2004)
この論文は米国の教科書Molecular Biology of the Cell, 5th edition(p929) と 6th edition (p855)、及び、Principles of Fluorescence Spectroscopy, 3rd edition(p23)と Faculty100にて紹介されました。
   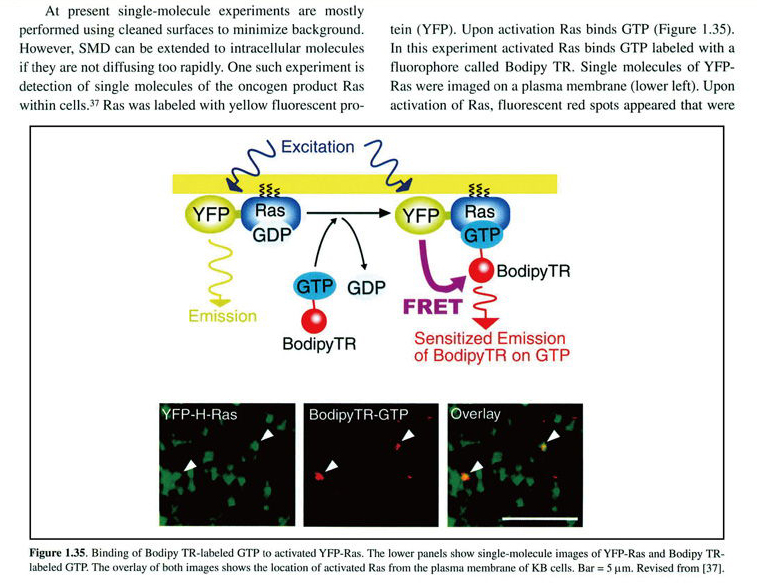
(Garland Science/Taylor & Francis LLCより転載許可を得ています)
-
Fujiwara T, Ritchie K, Murakoshi H, Jacobson K, & Kusumi A.
Phospholipids undergo hop diffusion in compartmentalized cell membrane.
J Cell Biol. 157, 1071–1081 (2002)
この論文は米国の教科書 Molecular Biology of the Cell, 5th (p648)と6th edition(p593)、及び、Lehninger Principles of Biochemistry, 4th & 5th & 6th (p398) editionにて紹介されました。
 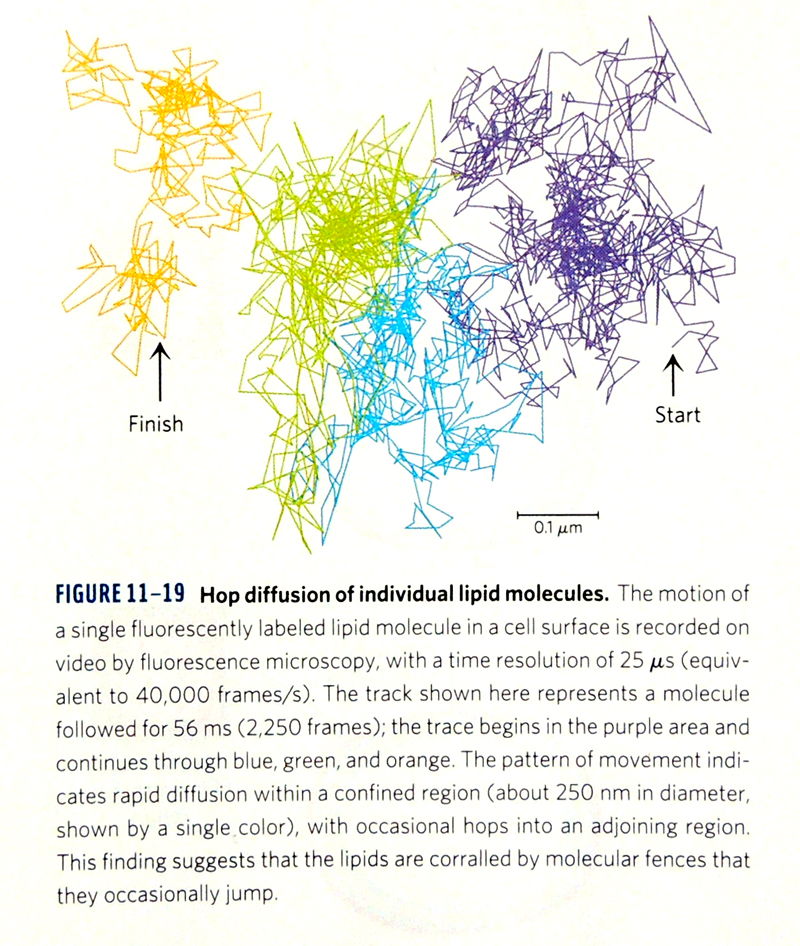  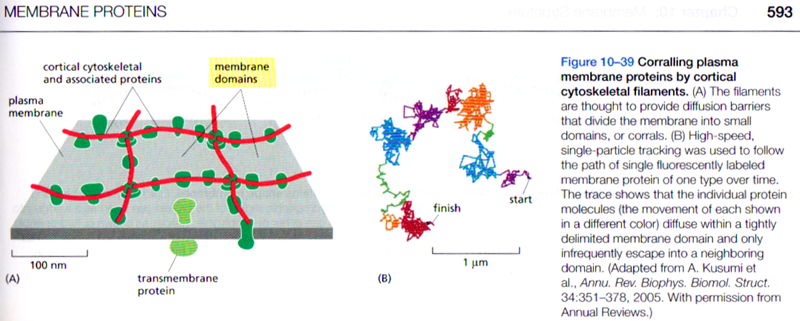
(Macmillan Educationより転載許可を得ています)
英語総説
-
Murakoshi H* and Shibata AC.
Optogenetic imaging of protein activity in the synapse by using 2-photon fluorescence lifetime imaging microscopy.
Optogenetics -Light-Sensing Proteins and Their Applications-
Springer-Verlag, Germany. p185–197 (2015)
-
Murakoshi H, Yasuda R.
Postsynaptic signaling during plasticity of dendritic spines.
Trends in Neurosci. 35, 135–143 (2012)
-
Yasuda R, Murakoshi H.
Themechanisms underlying the spatial spreading of signaling activity.
Curr. Opin. Neurobiol. 21, 313–321 (2011)
-
Kusumi A, Murakoshi H, Murase K, and Fujiwara T.
Single-molecule imaging of diffusion, recruitment, and activation of signaling molecules in living cells.
Biophysical Aspects of Transmembrane Signaling.
Springer-Verlag, Germany. 123–152 (2005)
-
Kusumi A, Nakada C, Ritchie K, Murase K, Suzuki K, Murakoshi H, Kasai RS, Kondo J, & Fujiwara T.
Paradigm Shift of the Plasma Membrane Concept from the Two-Dimensional Continuum Fluid to the Partitioned Fluid: High-Speed Single-Molecules Tracking of Membrane Molecules.
Annual Review of Biophysics and Biomolecular Structure. 34, 351–378 (2005)
日本語総説
-
村越秀治
「二光子蛍光寿命イメージング顕微鏡によるシグナル分子活性計測」
顕微鏡 2015年 第50巻 第2号 p106–110
-
村越秀治
シナプス内シグナル分子動態イメージング」
ブレインサイエンスレビュー2015 ブレインサイエンス振興財団 2015年 p191–210
- 村越秀治
「第三篇第1章6節 オプトジェネティクスによる神経細胞シナプス内シグナル伝達分子活性化イメージング」
オプトジェネティクス(光遺伝学)(株)エヌ・ティー・エス 2013年 p235–243
- 村越秀治
「二光子蛍光寿命イメージングによるRho GTPase活性化の単一シナプスレベル可視化解析」
生物物理 2012年 52(3) p158–159
- 村越秀治、安田涼平
「2光子蛍光寿命イメージング顕微鏡法を用いた単一シナプス内情報伝達の可視化」
生物物理 2010年 50(1) p23–26
- 村越秀治、安田涼平
「2光子蛍光寿命イメージング顕微鏡法を用いた単一シナプス内情報伝達の可視化」-生命現象の動的理解を目指すライブイメージング-
実験医学増刊 羊土社 2008年 p30–37
- 村越秀治
「第8章5節 低分子量Gタンパク質の活性化の1分子観察」 非侵襲・可視化技術ハンドブック-ナノ・バイオ・医療から情報システムまで-
(株)エヌ・ティー・エス 2007年
- 楠見明弘、村越秀治
「蛍光でわかるナノの世界:生体高分子を“見ながら”細胞の機能を探る」
ナノバイオロジー 共立出版 2004年 p9–22
- 村越秀治、楠見明弘
「Ras活性化の生細胞内1分子イメージング」
実験医学 羊土社 2004年 p2334–2335
- 村越秀治
「GTP結合によるRas活性化の1分子観察」
実験医学別冊バイオイメージングでここまでわかる 羊土社 2003年 p86–89
|
|










