1. Title
Deep brain stimulation driven by motor cortical activity can effectively treat Parkinson’s disease
2. Sub-headline
Researchers from University South Alabama College of Medicine, USA and National Institute for Physiological Sciences, Japan developed a new method of deep brain stimulation driven by motor cortical activity and showed its effectiveness in Parkinson’s disease monkeys to treat their motor symptoms.
3. Main text
Parkinson’s disease (PD) is a neurological disorder caused by progressive loss of dopaminergic neurons in the basal ganglia and characterized by tremor, movement difficulty, and muscle stiffness. PD affects 1% individual age over 60 years. Drug supplementation therapy compensates reduced dopamine and successfully treats PD symptoms. However, drug control of symptoms becomes difficult after a few years. For advanced PD, deep brain stimulation (DBS), applying electric stimulation to the subthalamic nucleus (STN), improves symptoms but associated with reduced efficacy with time and higher battery consumption. These problems prompted for a more efficient stimulation method.
In the team’s previous study, in PD monkeys, the movement signals originating from the primary motor cortex (M1) were blocked in the internal segment of the globus pallidus (GPi), causing movement difficulty. Manipulation of the STN unblocked the GPi and recovered movements. Therefore, the team speculated that controlling the DBS by the M1 activity might be a better treatment option for PD.
In this research, DBS was applied to the STN of the PD model monkeys. At first, the research team investigated the movement-related activity in the M1 and found that γ2 band (80-200 Hz) activity is a suitable biomarker for movements in PD condition. After that, recovery of upper limb reaching task was studied by applying DBS to the STN. They observed that the monkey could only reach out slowly without DBS whereas the application of conventional DBS with constant strength and frequency (cDBS) enabled smooth movement. Then, they controlled the strength and frequency of DBS by M1-γ2 activity and observed that this method, adaptive DBS (aDBS), enabled similarly smoother task. Moreover, aDBS battery consumption is 2/3 of the cDBS.
These results indicate that the newly developed aDBS enables modulation of stimulation parameters of STN-DBS in response to M1-γ2 band activity and is an extremely efficient treatment method in PD monkeys. In conclusion, M1-γ2 aDBS is an effective therapeutic approach which has a possible clinical application in PD patients and is expected to contribute to better DBS.
4. Figure and caption
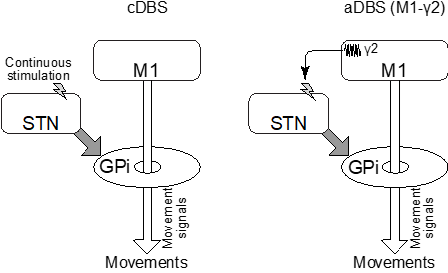
In Parkinson’s disease (PD), blockade of the movement signals originating from the primary motor cortex (M1) in the internal segment of the globus pallidus (GPi) causes movement difficulty. Conventional constant DBS (cDBS, left) applies electric stimulation continuously, unblocks the GPi, and enables movements. Newly developed adaptive DBS (aDBS, right) applies electric stimulation based on M1-γ2 band activity and unblocks the GPi when the movement signals reach the GPi.
5. Summary
Researchers from University South Alabama College of Medicine, USA and National Institute for Physiological Sciences, Japan developed a new adaptive deep brain stimulation (aDBS) driven by primary motor cortical activity. Both aDBS and conventional constant DBS (cDBS) significantly decreased reaction and movement times in Parkinson’s disease monkeys. The electric charge delivered with aDBS was lower than that with cDBS. aDBS is an effective therapeutic approach with lower power consumption.
Release Source


National Institute for Physiological Sciences
Japan Agency for Medical Research and Development
561


