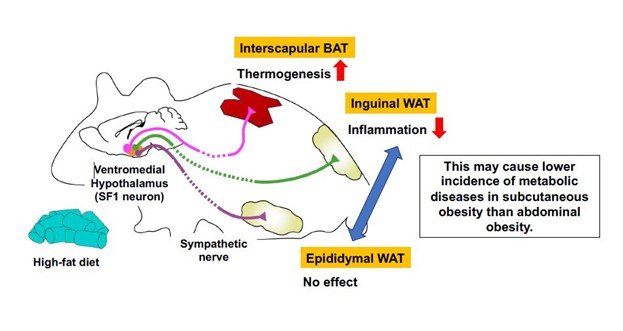
 SF1 neurons in the ventromedial hypothalamus were found to suppress inflammation in inguinal WAT in diet-induced obese mice through sympathetic nerves innervating the tissue, while they had no effect on epididymal WAT. The SF1 neurons also increased thermogenic function in interscapular BAT in diet-induced obese mice. Thus, the suppressive role of SF1 neurons in inflammatory response in subcutaneous (inguinal) WAT may explain why subcutaneous obesity has lower incidence of metabolic diseases than abdominal obesity.
SF1 neurons in the ventromedial hypothalamus were found to suppress inflammation in inguinal WAT in diet-induced obese mice through sympathetic nerves innervating the tissue, while they had no effect on epididymal WAT. The SF1 neurons also increased thermogenic function in interscapular BAT in diet-induced obese mice. Thus, the suppressive role of SF1 neurons in inflammatory response in subcutaneous (inguinal) WAT may explain why subcutaneous obesity has lower incidence of metabolic diseases than abdominal obesity.
Okazaki, Japan – Just as there are different types of fat in food, there are different types of fat tissue found within the body. White adipose tissue (WAT) is the most abundant form of fat, while brown adipose tissue (BAT) plays an important role in thermogenesis, the process in which heat is produced by burning calories. Recent studies indicate that differences in the association of WAT depot mass with cardiovascular disease may arise from different properties of subcutaneous versus abdominal WAT. Subcutaneous WAT manifests a lower extent of inflammation than that in abdominal WAT. Inflammation in those areas containing WAT can influence how obesity affects overall metabolic and cardiovascular health. However, it is unclear whether the nervous system controls inflammation differently in different regions of fat. Researchers in Japan have shed new light on a type of neuron in the brain that plays an important role in inflammation within these types of fat.
In a new study published in Cell Reports, researchers led by the National Institute for Physiological Sciences (NIPS) employed a high fat diet-fed mouse model to demonstrate how neurons that express a protein called steroidogenic factor 1 (SF1) influence inflammation in different regions or “depots” of fat.
It has been previously shown that SF1-expressing neurons reside in the hypothalamus, a region of the brain that controls hunger and peripheral metabolism. The NIPS-led research team set out to investigate the influence of SF1 neurons on different fat depots, including WAT found in the inguinal (subcutaneous) and epididymal (abdominal) regions, in mice who are fed a high-fat diet.
“We first investigated the effects of deactivating SF1-expressing neurons in the hypothalamus,” says senior author of the study Yasuhiko Minokoshi. “We found that mice without the SF1-expressing neuron gained substantially more weight on both normal and high-fat diets. Inguinal WAT weight increased in these mice, but interestingly, epididymal WAT weight actually decreased, while BAT showed no significant change. This indicated that SF1 neurons appear to regulate fat metabolism differently in different regions of the mouse body.”
The researchers found that expression of inflammatory and immune-cell markers and infiltration of the immune cells were elevated in the inguinal WAT of mice lacking SF1-expressing neurons and who were fed a high-fat diet, while in epididymal WAT, those markers exhibited high values in both mice lacking and not lacking SF1-expressing neurons. In BAT, markers of thermogenesis were significantly decreased in mice lacking SF1-expressing neurons. Conversely, activation of SF1 neurons in mice fed the high-fat diet led to decreased levels of inflammatory gene expression in inguinal WAT, but not in epididymal WAT, and increased thermogenic function in BAT.
“Our findings indicate that SF1 neurons in the hypothalamus help suppress inflammatory responses induced by a high-fat diet in the inguinal WAT,” says one of the double corresponding authors, Kunio Kondoh.
“These neurons also activate thermogenic activity in BAT, but do not appear to affect inflammatory response or thermogenesis in epididymal WAT,” says lead author Misbah Rashid.
The researchers further found that SF1 neurons exert their effect through sympathetic nerves that connect to inguinal WAT. Some specific SF1 neurons appear to regulate inflammatory response and thermogenesis differently in individual depots of fat through sympathetic nerves innervating the tissues. The team’s research improves our understanding of SF1 neurons and how they control inflammation, one of the driving factors of diseases related to obesity.
