The main goal of our research is to understand the regulation of neuronal circuit remodeling in development and recovery. In detail, we are focusing on the glial contribution to neuronal circuit function. We are trying to determine glial contribution to neuronal circuits in physiological and pathological conditions by visualizing fine structure, controlling activity, and recording specific synaptic transmissions in living animals using multi-photon microscopy. We focus on the relationship between neurons and astrocytes in the construction of pathological neuronal circuits in chronic pain. By leveraging this knowledge, we aim to manipulate astrocyte activity to drive the removal of such pathological circuits and restore normal sensation, with the ultimate goal of applying this in a clinical setting. We also study experience-dependent remodeling in sensory neuronal circuits during development. We use behavioral analysis, in vivo imaging, and in vitro electrophysiology to clarify the correlation between the development of behavioral patterns and its basis in synaptic plasticity changes.
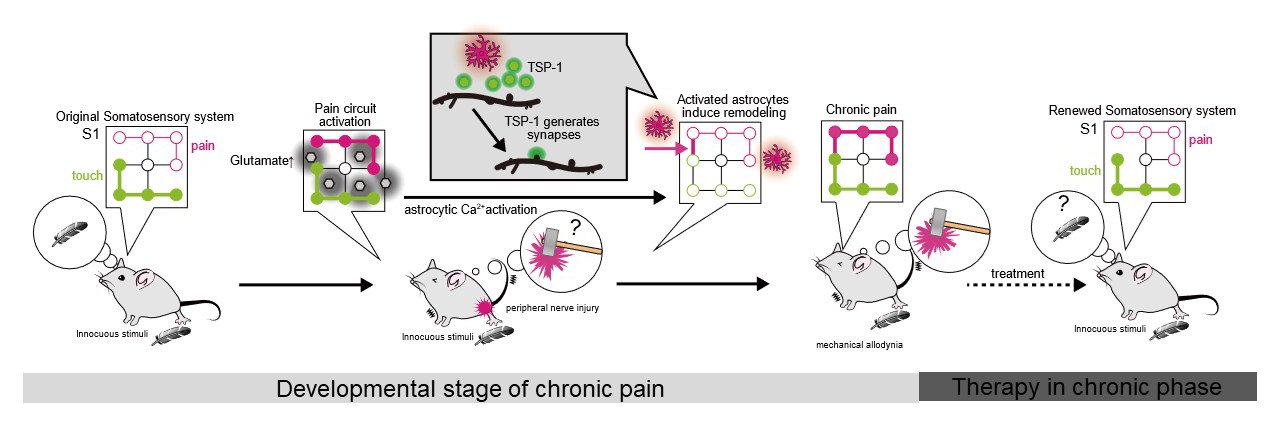
The progress of technology has brought about a breakthrough in life science. We recently revealed neural activity-dependent pH changes in the living brain with single-cell level resolution using a CMOS image sensor which we had newly developed. We observed that pH changes propagated throughout the epileptic hippocampus prior to the occurrence of measurable electrical epileptic activity. Based on this, we have started a collaboration with medical clinics and industry to develop new tools for predicting the onset of epileptic seizures in human patients.
Fig1. CMOS-based ion image sensor revealed neuronal activity-dependent pH changes in the living brain
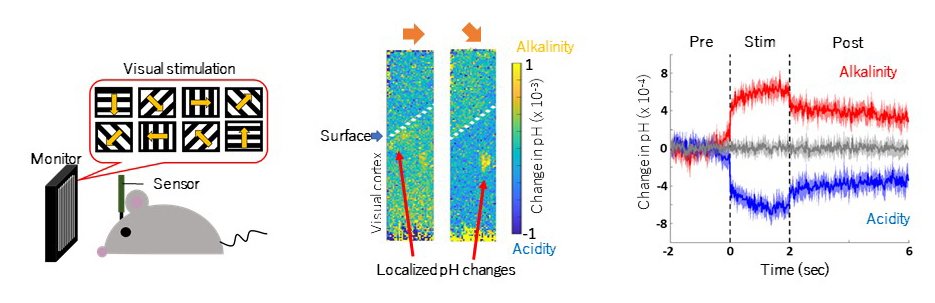
Fig2. Pain circuit reorganization with activated astrocytes as a therapeutic approach